Arch Iran Med. 25(4):250-256.
doi: 10.34172/aim.2022.41
Original Article
Immunohistochemical Overexpression of Cyclin D1 in Tunisian Invasive Breast Carcinoma Women
Lobna Bouzidi 1, 2, *  , Saadia Makni 1, 2, Jihen Feki 3, Rim Kallel 1, 2, Soumaya Graja 1, 2, Naourez Gouiaa 1, 2, Tahya Sellami-Boudawara 1, 2, Manel Mellouli 1, 2
, Saadia Makni 1, 2, Jihen Feki 3, Rim Kallel 1, 2, Soumaya Graja 1, 2, Naourez Gouiaa 1, 2, Tahya Sellami-Boudawara 1, 2, Manel Mellouli 1, 2
Author information:
1Department of Pathology and Research laboratory LR18SP10, University Hospital Habib Bourguiba, Sfax, Tunisia
2Medical School of Sfax, University of Sfax, Sfax, Tunisia
3Department of Medical Oncology, University Hospital Habib Bourguiba, Sfax, Tunisia
*
Corresponding Author: Lobna Bouzidi, MD; Department of pathology Habib Bourguiba University Hospital. El Ain Road Km 0.5 3029. Sfax-Tunisia. Phone:+21644876050; E-mail:
lobnalobna1992@gmail.com
Abstract
Background:
Breast cancer represents the most frequent cancer and cause of death in women worldwide and in Tunisia. Cyclin D1 is a gene of cell cycle regulation. It represents a potential oncogene in invasive breast cancer; however; the results are conflicting. We performed a retrospective study aiming to analyze the prognostic impact of cyclin D1 expression in patients with invasive breast carcinoma of no special type and its relation with clinical-pathological features.
Methods:
One hundred cases of invasive breast carcinoma of no special type diagnosed between 2009 and 2011 were included in this study. Immunohistochemical (IHC) staining was performed for cyclin D1 in all cases. Results were analyzed statistically.
Results:
Cyclin D1 positivity was seen in 74 cases (74%), of which 32 cases (32%) showed strong immunoreactivity. Cyclin D1 staining was statistically significantly associated with estrogen receptor (ER) and progesterone receptor (PR) positivity (P<0.0001) and with low grade SBR (P=0.007). None of the clinical data and other pathological features had any association with cyclin D1 expression (P>0.05). Univariate analysis revealed that expression of cyclin D1 was not statistically associated with overall survival (OS) and disease-free survival (DFS) (P=0.459 and P=0.564, respectively).
Conclusion:
These results confirm that cyclin D1 overexpression can be employed as a beneficial prognostic marker and suggest that anti-cyclin D1 therapy may be efficient, especially for ER positive tumors.
Keywords: Breast cancer, Cyclin D1, Estrogen receptor, Progesterone receptor, Prognosis
Copyright and License Information
© 2022 The Author(s).
This is an open-access article distributed under the terms of the Creative Commons Attribution License (
https://creativecommons.org/licenses/by/4.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Cite this article as: Bouzidi L, Makni S, Feki J, Kallel R, Graja S, Gouiaa N, et al. Immunohistochemical overexpression of cyclin D1 in tunisian invasive breast carcinoma women. Arch Iran Med. 2022;25(4):250-256. doi: 10.34172/aim.2022.41
Introduction
Breast cancer represents the most frequent cancer and cause of death in women worldwide and in Tunisia. Its mortality rate is 6.6%.1 This cancer represents a heterogeneous disease which includes several pathological and molecular subtypes showing different and unpredictable outcomes and treatment responses.2
Classical and well-established clinical and pathological prognostic markers like progesterone receptor (PR), estrogen receptor (ER) and human epidermal growth factor 2 (HER2) are necessary and useful but they do not always predict the outcomes of these tumors.3-5
The provision of new prognostic factors is crucial for better understanding of the molecular mechanism of carcinogenesis, which allows improving the outcome and supplying new therapeutic perspectives.6
Several recent studies addressing cyclin D1 have shown the role of this protein in the development of a substantial proportion of tumors, including breast cancer, as well as the therapeutic management.7,8
Cyclin D1 plays a dual role as one of the main proteins of cell cycle regulation and as a transcriptional factor.9 It is encoded by the gene, oncogenic c-terminal cyclin D1 (CCND1) or parathyroid neoplasia gene (PRAD1), located on chromosome 11q13.9 The deletion of cyclin D1 causes poor mammary gland development and delivered protection from the development of breast cancer.10 On the other hand, cyclin D1 overexpression leads to excessive mammary proliferation and thus, high incidence of BC.11 Indeed, it allows progression through G1-S phase by binding to cyclin-dependent kinase 4 (CDK4) and cyclin-dependent kinase 6 (CDK6).12
However, the role of cyclin D1 overexpression in the pathogenesis and prognosis of breast cancer remains controversial; researchers have reported inconsistent and conflicting results.6
The goals of our study were to estimate the expression of cyclin D1 in invasive breast carcinoma, to evaluate the relation of this expression with other clinical-pathological prognostic factors and to judge the prognostic significance of cyclin D1 expression.
Materials and Methods
Clinical and Pathological Data
This is a retrospective study of female patients presenting with breast carcinoma between 2009 and 2011. Patients originated from southern Tunisia. Surgical resection was performed for all patients at the Department of Gynecology and Obstetrics of the Hedi Chaker University Hospital (Sfax, Tunisia). The specimens (n = 100) were formalin-fixed and paraffin-embedded at the Department of Pathology of the Habib Bourguiba University Hospital (Sfax, Tunisia). Patients who received radiation before surgical resection or neoadjuvant chemotherapy were not included. We used Microsoft Excel version 7.0 and medical files archived at the department of Pathology of the Habib Bourguiba University Hospital (Sfax, Tunisia) to collect pathological data. Hematoxylin and eosin slides were also retrieved from the department of Pathology of the Habib Bourguiba University Hospital (Sfax, Tunisia).
For each specimen, the following pathological data were collected: tumor multifocality, tumor size, histological grade [according to Elston-Ellis modification of Scarff-Bloom-Richardson system (SBR)],13 presence of lymphovascular invasion (LVI), presence of perineural invasion (PNI), tumor necrosis, concomitant carcinoma in situ (CIS), Paget disease, surgical margin status, ER and PR status,14 HER2 status15 and proliferation index Ki-67 (considered overexpressed if ≥ 20%).16 HER2 staining was analyzed according to the Wolff criteria.15 Tumors were considered positive for HER2 if immunostaining was scored as 3 + . HER2 cases evaluated as 2 + were examined with fluorescent in situ hybridization (FISH). ER and PR were considered positive when more than 1% of the infiltrating tumor cell nuclei were marked.14
We identified five molecular subtypes: Luminal A (LA) if ER and/or PR positive, HER2 negative and Ki-67 < 20%; Luminal B like (LB-Like) if ER and/or PR positive, HER2 negative and Ki- 67 > 20%; Luminal B (LB) if ER and/or PR positive and HER2 positive; HER2-positive breast cancer (HER2 +) if both ER and PR negative and HER2 positive and finally, Triple Negative Breast Cancer (TNBC) if HR and HER2 negative.17
The pathological TNM classification and staging was done as per American Joint Committee on Cancer (AJCC) guidelines.18
Clinical data were collected from medical records at the department of Oncology of the Habib Bourguiba University Hospital (Sfax, Tunisia) and they included age, menopausal status, distant metastasis at diagnosis and outcomes. Overall survival (OS) was designated as time from primary surgical treatment to the date of the last follow-up or death. Disease-free-survival (DFS) was designated as time during which no sign of cancer appeared after treatment.
Immunohistochemical Staining for Cyclin D1
The expression of cyclin D1 (clone E1544; dilution 1:50; Spring) was evaluated by immunohistochemistry (IHC). We used 3 μm slices, dried overnight at 40°C and deparaffinized in xylene. Later, slices were rehydrated in alcohol at 100°C then at 95°C and washed in purified water.
Antigen retrieval was evaluated using a boiled water bath with basic buffer (pH 9) for 40 minutes until the temperature climbed to 98°C. They were then allowed to cool spontaneously.
The endogenous peroxidase activity was neutralized by hydrogen peroxide (H2O2, 3%) for 10 min. The sections were washed with distilled water and with phosphate buffered saline (PBS). The sections were then covered with antibody for cyclin D1 at 1:30 dilution for 1 hour. Next, sections were incubated with biotin-conjugated secondary antibody for 20 min and then incubated using streptavidin biotin system for 20 minutes at room temperature. PBS washing for 5 minutes was performed for each step. The reactions became clear after the immersion of sections in 3, 3 diaminobenzidine a substrate–chromogen solution for 20 minutes. The final step was to counterstain the slides with Mayer hematoxylin, to mount them permanently, and to examine them with a standard light microscope.
Immunostaining for cyclin D1 was interpreted by two pathologists. It was considered positive when at least 10% or more of the tumor cells showed nuclear expression regardless of the intensity of staining.2
Statistical Analyses
Data were analyzed using the SPSS software (version 20.0). The correlation between cyclin D1 expression and the clinical-pathological factors was evaluated by the Chi-square and the Fisher exact tests.
Survival analysis was performed using the Kaplan-Meier method and compared by the log-rank test. A Cox regression model was used to describe the risk factors with survival parameters. A P value ≤ 0.05 showed statistical significance.
Results
Population Characteristics
Clinical and pathological data arelistedin Table 1. Paget disease was noted in 6 % of cases. Surgical margins were positive in 12 cases. The median follow-up was 53, 3 months. The 5-year OS rate was 82.1%. The 5-year DFS rate was 80.1%.
Table 1.
Relationship between Cyclin D1 Expression and Clinical-Pathological Characteristics in Breast Cancer
|
Variable
|
Cyclin D1 Expression
|
|
<10% (n=26)
|
≥10 (n=74)
|
P
|
| Age (y) (mean = 50.5 years) |
≤ 45 (n = 35) |
8 (22.9%) |
27 (77.1%) |
0.599 |
| > 45 (n = 65) |
18 (27.7%) |
47 (72.3%) |
| Hormonal status |
Menopausal (n = 45) |
11 (24.5%) |
34 (75.5%) |
0.748 |
| Not menopausal (n = 55) |
15 (27.3%) |
40 (72.7%) |
| Multifocality |
Yes (n = 17) |
2 (11.8%) |
15 (88.2%) |
0.225 |
| No (n = 83) |
24 (29%) |
59 (71%) |
| Tumor size (cm) |
≤ 2 (n = 23) |
6 (73.9%) |
17 (26.1%) |
0.7 |
| 2-5 (n = 62) |
47 (75.8%) |
15 (24.2%) |
| > 5 (n = 15) |
5 (33.3%) |
10 (66.7%) |
| SBR Grading |
Grade I (n = 16) |
4 (25%) |
12 (75%) |
0.8 |
| Grade II (n = 61) |
17 (27.9%) |
44 (72.1%) |
| Grade III (n = 23) |
5 (21.7%) |
18 (78.3%) |
| Low Grade (Grade I,II) (n = 77) |
21 (27.3%) |
56 (72.7%) |
0.007
|
| High Grade (Grade III) (n = 23) |
5 (21.7%) |
18 (78.3%) |
| LVI |
Yes (n = 56) |
17 (30.3%) |
39 (69.7%) |
0.262 |
| No (n = 44) |
9 (20.5%) |
35 (79.5%) |
| PNI |
Yes (n = 27) |
4 (14.8%) |
23 (85.2%) |
0.121 |
| No (n = 73) |
22 (30.1% |
51 (69.9%) |
| Tumor necrosis |
Yes (n = 18) |
8 (44.4%) |
10 (55.6%) |
0.073 |
| No (n = 82) |
18 (22%) |
64 (78%) |
| CIS |
Yes (n = 81) |
19 (23.5%) |
62 (76.5%) |
0.253 |
| No (n = 19) |
7 (36.8%) |
12 (63.2%) |
| HER-2 expression status |
Amplified (n = 28) |
6 (21.4%) |
22 (78.6%) |
0.516 |
| Non amplified (n = 72) |
20 (27.8%) |
52 (72.2%) |
| ER expression status |
Positive (n = 74) |
12 (16.2%) |
62 (83.8%) |
<0.0001
|
| Negative (n = 26) |
14 (53.8%) |
12 (46.2%) |
| PR expression status |
Positive (n = 63) |
9 (14.3%) |
54 (85.7%) |
<0.0001
|
| Negative (n = 37) |
17 (46%) |
20 (54%) |
| Ki67 |
≥ 20% (n = 50) |
14 (28%) |
36 (72%) |
0.648 |
| < 20% (n = 50) |
12 (24%) |
38 (76%) |
| Molecular subtype |
LA (n = 20) |
9 (45%) |
11 (55%) |
0.2 |
| LB- HER2 negative (n = 36) |
8 (22.2%) |
28 (77.8%) |
| LB-HER2 positive (n = 18) |
5 (27.8%) |
13 (72.2%) |
| HER2 + (n = 11) |
2 (18.2%) |
9 (81.8%) |
| TNBC (n = 15) |
2 (86.7%) |
13 (13.3%) |
| pT status |
p T1 (n = 23) |
5 (21.7%) |
18 (78.3%) |
0.1 |
| p T2 (n = 54) |
12 (22.2%) |
42 (77.8%) |
| p T3 (n = 14) |
4 (28.6%) |
10 (71.4%) |
| p T4 (n = 9) |
5 (55.6%) |
4 (44.4%) |
| Lymph node status |
N0 (n = 42) |
8 (19%) |
34 (81%) |
0.3 |
| N1 (n = 32) |
12 (37.5%) |
20 (62.5%) |
| N2 (n = 15) |
4 (26.7%) |
11 (73.3%) |
| N3 (n = 11) |
2 (18.2%) |
9 (81.8%) |
| Metastasis |
M + (n = 8) |
1 (12.5%) |
7 (87.5%) |
0.676 |
| M – (n = 92) |
25 (27.2%) |
67 (72.8%) |
SBR, Scarff-Bloom-Richardson; LVI, Lymphovascular invasion; PNI, Perineural invasion; Her-2, Human Epidermal Growth 2; ER, estrogen receptor; PR, progesterone receptor; LA, luminal A; LB, Luminal B; TNBC, triple negative breast cancer
Bold highlighted red values show significance (P≤ 0.05).
Cyclin D1 Expression and Relation with other Clinical-pathological Factors
Cyclin D1 was found to be positive in 74 cases, 32 of which showed strong immunoreactivity and 14 showed weak immunoreactivity (Figure 1).
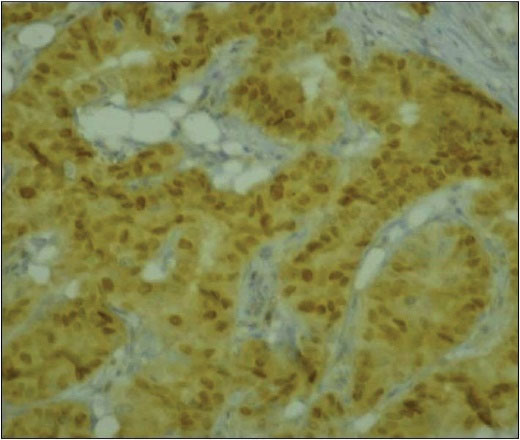
Figure 1.
Cyclin D1 Immunostaining. Strong nuclear cyclin D1 staining (IHC × 200).
.
Cyclin D1 Immunostaining. Strong nuclear cyclin D1 staining (IHC × 200).
The relationbetween cyclin D1 expression and clinical-pathological factors is shown in Table 1. Cyclin D1 staining was significantly correlated with ER and PR positivity (P< 0.0001), with low grade SBR (P = 0.007).
Most cases of positive cyclin D1 showed absence of tumor necrosis (P= 0.073). Moreover, none of the clinical data and other pathological features showed any association with cyclin D1 expression (Table 1).
Univariate analysis showedthat expression of cyclin D1 was not statistically correlatedwith OS and DFS. The 5-year OS rate was 81.2% in tumors with positive staining for cyclin D1 and 84.2% in tumors without cyclin D1 expression (P= 0.459) (Figure 2A). Similarly, the expression of cyclin D1 was not associated with better DFS (P= 0.564) (Figure 2B).
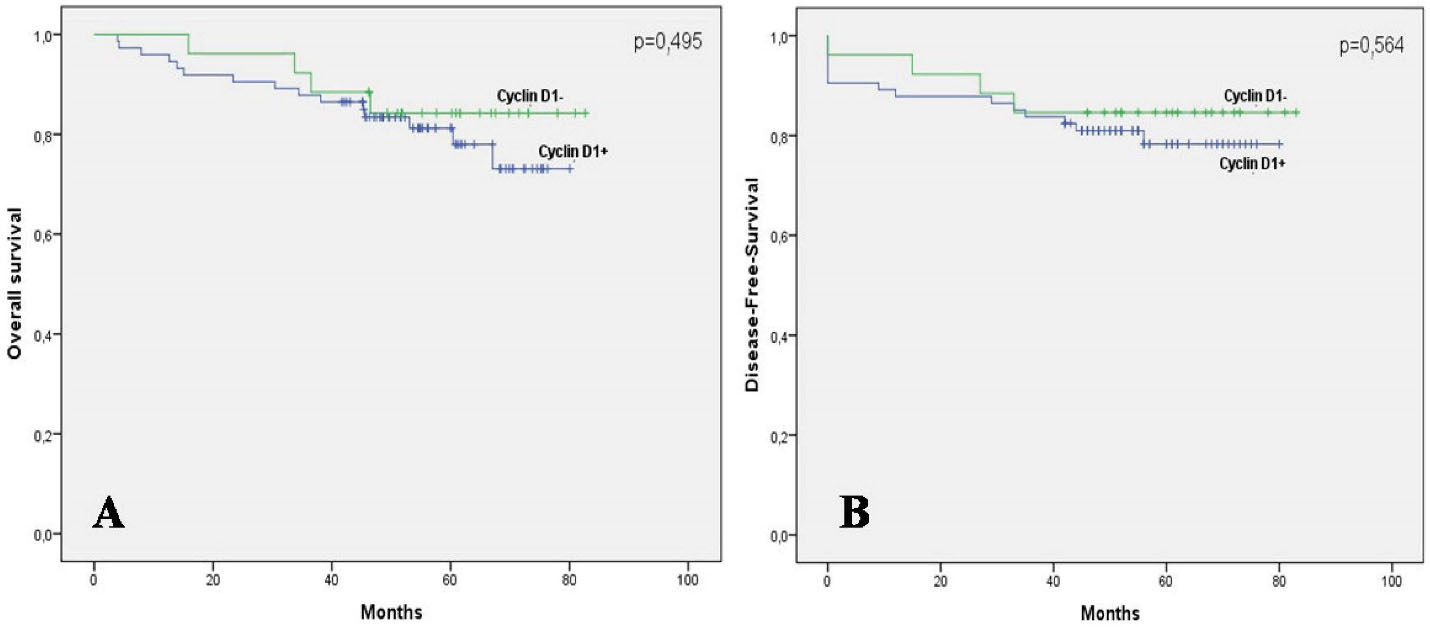
Figure 2.
Survival Outcomes According to Cyclin D1 Expression. (A) Kaplan-Meier curves of overall survival according to Cyclin D1 expression: the 5-year OS rate was 81.2% in tumors with positive staining for Cyclin D1 and 84.2% in tumors without Cyclin D1 expression (p = 0.459). (B) Kaplan-Meier curves of Disease-Free-survival according to Cyclin D1 expression: the expression of Cyclin D1 was not associated with better DFS (P = 0.564).
.
Survival Outcomes According to Cyclin D1 Expression. (A) Kaplan-Meier curves of overall survival according to Cyclin D1 expression: the 5-year OS rate was 81.2% in tumors with positive staining for Cyclin D1 and 84.2% in tumors without Cyclin D1 expression (p = 0.459). (B) Kaplan-Meier curves of Disease-Free-survival according to Cyclin D1 expression: the expression of Cyclin D1 was not associated with better DFS (P = 0.564).
A multivariate Cox model was performed to evaluate the risk factorsassociated with OS and DFS. Only factors which were a significant prognostic factor in the univariate study were included in the model (Tumor size, SBR grade, LVI, pT status, pN status and metastasis). The results are presented in Table 2 and Table 3.
Table 2.
Results of Multivariate Cox Regression Model for Overall Survival
|
Variables
|
Coefficient
|
Errer
Standard
|
P
Value
|
HR
|
95% CI
|
|
Lower
|
Upper
|
| Tumor size |
1.312 |
0.883 |
0.137 |
3.712 |
0.658 |
20.932 |
| SBR Grade |
1.514 |
0.672 |
0.024
|
4.546 |
1.219 |
16.956 |
| LVI |
12.668 |
388.107 |
0.974 |
1.52 |
0.000 |
0 |
| pT status |
0.014 |
0.484 |
0.978 |
1.014 |
0.393 |
2.616 |
| pN status |
0.459 |
0.389 |
0.238 |
1.583 |
0.739 |
3.391 |
| Metastasis |
4.418 |
1.252 |
0.000
|
82.909 |
7.127 |
964.494 |
HR, hazard ratio; 95% CI, 95% confidence interval; SBR, Scarff-Bloom-Richardson; LVI, lymphovascular invasion.
Bold highlighted values show significance (P ≤ 0.05).
Table 3.
Results of Multivariate Cox Regression Model for Disease Free Survival
|
Variables
|
Coefficient
|
Errer Standard
|
P
Value
|
HR
|
95% CI
|
|
Lower
|
Upper
|
| Tumor size |
0.824 |
0.775 |
0.288 |
2.281 |
0.499 |
10.423 |
| SBR Grade |
1.215 |
0.594 |
0.041
|
3.371 |
1.053 |
10.796 |
| LVI |
12.224 |
283.428 |
0.966 |
1.103 |
0.000 |
0 |
| pT status |
0.423 |
0.384 |
0.270 |
1.527 |
0.720 |
3.239 |
| pN status |
0.554 |
0.364 |
0.129 |
1.740 |
0.852 |
3.552 |
HR, hazard ratio; 95% CI, 95% confidence interval; SBR, Scarff-Bloom-Richardson; LVI, lymphovascular invasion.
Bold highlighted values show significance (P ≤ 0.05).
Discussion
Immunohistochemical Overexpression of Cyclin D1
Overexpression of cyclin D1 varies from 23% to 81.4% according to studies published in the English literature.19,20 In our study, overexpression of cyclin D1 was noted in 74% of cases.
This large difference between different series may be explained by several factors. Indeed, immunohistochemistry is performed using different clones of cyclin D1 which require specific protocol for each one and thus influence cyclin D1 staining. Next, this large discrepancy may also be influenced by the intrinsic characteristics of tumors.
The overexpression of cyclin D1 may be detected by immunohistochemistry, using antibody against cyclin D1, even without any apparent increase in the copy numbers of CCND1.21
Association of Cyclin D1 Expression with Clinical-Pathological Data
The majority of studies have shown a significant association between overexpression of cyclin D1 and good prognostic parameters such as small tumor size and good tumor differentiation with low grade SBR.7,22,23 A significant correlation between expression of cyclin D1 and low tumor grade has been reported by many authors, which is in line with our results.23-25
Indeed, cyclin D1 affects cell maturation and differentiation; high expression of cyclin D1 suppresses deoxyribonucleic acid (DNA) replication by linking to the proliferating cell nuclear antigen and to cyclin-dependent kinase 2 (CDK2). 6
A significant association has been reported in almost all publications between cyclin D1 overexpression and breast cancer subtypes ER-positive.2,7,8,22,26-31
The expression of cyclin D1 was significantly correlated with PR and ER positivity in the present study (P < 0.0001).2,9,23,32
In a study by Huang et al. on 101 cases of breast invasive carcinoma of no specific type, the overexpression of cyclin D1 was statistically correlated with early tumor stage (Stage I or II) (P = 0.047); 85% of these tumors were positive for ER (P < 0.0001) and 78.75% were positive for PR (P = 0.001).7
Likewise, Mylona et al demonstrated that overexpression of cyclin D1 was associated with small tumors (P = 0.017), ER + (P < 0.0001), RP + (P < 0.0001), low grade SBR (P < 0.0001) and with low expression of protein 53 (p53) (P < 0.001).33
In fact, estrogen and progesterone increase the transcription of the CCND1 gene, thus leading to an overexpression of the cyclin D1 protein.34 So, Luminal A or B tumors express more cyclin D1 whereas tumors with basal-like phenotype lose this expression.7,31
Studies showed that cyclin D1 acts like a transcriptional factor without any interaction with its associated CDKs.35 Somehow, it can bind to the hormone binding domain of the ER, thus activating the mediators and the transcriptional regulators of the ER.10,36 Simultaneously, this estrogen-cyclin D1 linkage acts by enhancing the PR expression through a novel estrogen- and cyclin D1-responsive enhancer in the PR gene.35 Consequently, cyclin D1 has a pivotal role in augmenting estrogen and progesterone effects in the mammary gland.35
This CDK-independent cyclin D1 function leads to poor response to anti-estrogen treatment by acquiring an agonist effect.36 In fact, Kenny et al37 and Stendahl et al38 reported that overexpression of cyclin D1 is a predictor of resistance to hormonotherapy based on tamoxifen.
Its prognostic value in terms of survival has been a subject of controversy; indeed, many authors have shown that cyclin D1 overexpression in breast cancer is correlated with a good outcome like Chung et al. who found that the 5-year-overall survival increased from 89.9% for cyclin D1 overexpression tumors to 98.9% for cyclin D1 negative tumors (P= 0.008).20 In this same study, the overexpression of cyclin D1 was correlated with a longer survival rate after tumor recurrence – 61 months for tumor overexpressing cyclin D1 versus 26 months for tumor with negative cyclin D1 (P= 0.012).20 Indeed, some authors reported that overexpression of cyclin D1 showed a statistically significant association with low proliferative rate.24,25 Thus, cyclin D1 overexpression could be a marker of a less indolent progression of the disease after tumor recurrence.20
On the other hand, some authors have not shown a significant association in terms of OS or DFS which is consistent with our results.25,28,39
However, some authors have reported that deletion of cyclin D1 can play a protector role, thus protecting against the development of breast carcinoma.10,40 In fact, cyclin D1 overexpression can be a predictor of poor prognosis.37,41,42 Ahlin et al found that cyclin D1 overexpression was associated with a high proliferative index and an increased risk of mortality in patients with ER positive breast cancer but not in those with ER negative breast cancer.43
Given these findings, the relationship between cyclin D1 overexpression and hormone receptors in breast cancer seems to be well established among authors. Researches about new prognostic markers in breast cancer are currently in progress and provide a huge amount of information in terms of better understanding the tumor microenvironment44 and the gene signature.45 However, the prognostic value of cyclin D1 overexpression in breast cancer is controversial and needs further investigations.
Actually, cyclin D1 is arising as an important prognostic parameter in invasive breast cancer, but it is not employed yet in routine practice. Its substantial role in predicting treatment response to tamoxifen should be considered to improve the management of ER-positive tumors.
It constitutes a novel therapeutic target especially in patients with ER positive tumors to overcome resistance to tamoxifen.
Acknowledgements
We thank all clinicians and technicians of our departments for technical support.
Authors’ Contribution
LB and SM assure the integrity of the study. LB and SM: Conceptualization, formal analysis and data curation. LB, SM and SG: Investigation; JF and NG: Methodology; RK, TSB and MM: Supervision and validation.
Conflict of Interest Disclosures
The authors declare that they have no conflict of interest
Ethical Statement
Authors followed the instruction of the Committee on Publication Ethics (COPE).
References
- Bray F, Ferlay J, Soerjomataram I, Siegel RL, Torre LA, Jemal A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin 2018; 68(6):394-424. doi: 10.3322/caac.21492 [Crossref] [ Google Scholar]
- Ravikumar G, Ananthamurthy A. Cyclin D1 expression in ductal carcinoma of the breast and its correlation with other prognostic parameters. J Cancer Res Ther 2014; 10(3):671-5. doi: 10.4103/0973-1482.138135 [Crossref] [ Google Scholar]
- Galant C, Berlière M, Leconte I, Marbaix E. Overview of histopathological prognostic factors in breast cancer, including newcomers. Imagerie de la Femme 2010; 20(1):9-17. doi: 10.1016/j.femme.2010.02.002 [Crossref] [ Google Scholar]
- Dialla PO, Dabakuyo TS, Marilier S, Gentil J, Roignot P, Darut-Jouve A. Population-based study of breast cancer in older women: prognostic factors of relative survival and predictors of treatment. BMC Cancer 2012; 12:472. doi: 10.1186/1471-2407-12-472 [Crossref] [ Google Scholar]
- Dabakuyo TS, Bonnetain F, Roignot P, Poillot ML, Chaplain G, Altwegg T. Population-based study of breast cancer survival in Cote d’Or (France): prognostic factors and relative survival. Ann Oncol 2008; 19(2):276-83. doi: 10.1093/annonc/mdm491 [Crossref] [ Google Scholar]
- Mohammadizadeh F, Hani M, Ranaee M, Bagheri M. Role of cyclin D1 in breast carcinoma. J Res Med Sci 2013; 18(12):1021-5. [ Google Scholar]
- Huang W, Nie W, Zhang W, Wang Y, Zhu A, Guan X. The expression status of TRX, AR, and cyclin D1 correlates with clinicopathological characteristics and ER status in breast cancer. Onco Targets Ther 2016; 9:4377-85. doi: 10.2147/ott.s94703 [Crossref] [ Google Scholar]
- Guo L, Liu S, Jakulin A, Yilamu D, Wang B, Yan J. Positive expression of cyclin D1 is an indicator for the evaluation of the prognosis of breast cancer. Int J Clin Exp Med 2015; 8(10):18656-64. [ Google Scholar]
- Lengare PV, Sinai Khandeparkar SG, Joshi AR, Gogate BP, Solanke SG, Gore SH. Immunohistochemical expression of cyclin D1 in invasive breast carcinoma and its correlation with clinicopathological parameters. Indian J Pathol Microbiol 2020; 63(3):376-81. doi: 10.4103/ijpm.ijpm_106_20 [Crossref] [ Google Scholar]
- Sutherland RL, Musgrove EA. Cyclin D1 and mammary carcinoma: new insights from transgenic mouse models. Breast Cancer Res 2002; 4(1):14-7. doi: 10.1186/bcr411 [Crossref] [ Google Scholar]
- Garrido-Castro AC, Goel S. CDK4/6 inhibition in breast cancer: mechanisms of response and treatment failure. Curr Breast Cancer Rep 2017; 9(1):26-33. doi: 10.1007/s12609-017-0232-0 [Crossref] [ Google Scholar]
- Hwang TS, Han HS, Hong YC, Lee HJ, Paik NS. Prognostic value of combined analysis of cyclin D1 and estrogen receptor status in breast cancer patients. Pathol Int 2003; 53(2):74-80. doi: 10.1046/j.1440-1827.2003.01441.x [Crossref] [ Google Scholar]
- Elston CW, Ellis IO. Pathological prognostic factors in breast cancer. I. The value of histological grade in breast cancer: experience from a large study with long-term follow-up. Histopathology 1991; 19(5):403-10. doi: 10.1111/j.1365-2559.1991.tb00229.x [Crossref] [ Google Scholar]
- Hammond ME, Hayes DF, Wolff AC, Mangu PB, Temin S. American Society of Clinical Oncology/College of American Pathologists guideline recommendations for immunohistochemical testing of estrogen and progesterone receptors in breast cancer. J Oncol Pract 2010; 6(4):195-7. doi: 10.1200/jop.777003 [Crossref] [ Google Scholar]
- Wolff AC, Hammond ME, Hicks DG, Dowsett M, McShane LM, Allison KH. Recommendations for human epidermal growth factor receptor 2 testing in breast cancer: American Society of Clinical Oncology/College of American Pathologists clinical practice guideline update. Arch Pathol Lab Med 2014; 138(2):241-56. doi: 10.5858/arpa.2013-0953-SA [Crossref] [ Google Scholar]
- Dowsett M, Nielsen TO, A’Hern R, Bartlett J, Coombes RC, Cuzick J. Assessment of Ki67 in breast cancer: recommendations from the International Ki67 in Breast Cancer working group. J Natl Cancer Inst 2011; 103(22):1656-64. doi: 10.1093/jnci/djr393 [Crossref] [ Google Scholar]
- Franchet C, Duprez-Paumier R, Lacroix-Triki M. [Molecular taxonomy of luminal breast cancer in 2015]. Bull Cancer 2015; 102(6 Suppl 1):S34-46. doi: 10.1016/s0007-4551(15)31216-9 [Crossref] [ Google Scholar]
- Giuliano AE, Edge SB, Hortobagyi GN. Eighth edition of the AJCC cancer staging manual: breast cancer. Ann Surg Oncol 2018; 25(7):1783-5. doi: 10.1245/s10434-018-6486-6 [Crossref] [ Google Scholar]
- Takano Y, Takenaka H, Kato Y, Masuda M, Mikami T, Saegusa M. Cyclin D1 overexpression in invasive breast cancers: correlation with cyclin-dependent kinase 4 and oestrogen receptor overexpression, and lack of correlation with mitotic activity. J Cancer Res Clin Oncol 1999; 125(8-9):505-12. doi: 10.1007/s004320050309 [Crossref] [ Google Scholar]
- Chung J, Noh H, Park KH, Choi E, Han A. Longer survival in patients with breast cancer with cyclin d1 over-expression after tumor recurrence: longer, but occupied with disease. J Breast Cancer 2014; 17(1):47-53. doi: 10.4048/jbc.2014.17.1.47 [Crossref] [ Google Scholar]
- Gillett C, Fantl V, Smith R, Fisher C, Bartek J, Dickson C. Amplification and overexpression of cyclin D1 in breast cancer detected by immunohistochemical staining. Cancer Res 1994; 54(7):1812-7. [ Google Scholar]
- Naidu R, Wahab NA, Yadav MM, Kutty MK. Expression and amplification of cyclin D1 in primary breast carcinomas: relationship with histopathological types and clinico-pathological parameters. Oncol Rep 2002; 9(2):409-16. [ Google Scholar]
- Sarkar S, Kanoi A, Bain J, Gayen R, Das KN. Correlation between cyclin D1 expression and standard clinicopathological variables in invasive breast cancer in Eastern India. South Asian J Cancer 2015; 4(4):155-9. doi: 10.4103/2278-330x.175950 [Crossref] [ Google Scholar]
- Peurala E, Koivunen P, Haapasaari KM, Bloigu R, Jukkola-Vuorinen A. The prognostic significance and value of cyclin D1, CDK4 and p16 in human breast cancer. Breast Cancer Res 2013; 15(1):R5. doi: 10.1186/bcr3376 [Crossref] [ Google Scholar]
- van Diest PJ, Michalides RJ, Jannink L, van der Valk P, Peterse HL, de Jong JS. Cyclin D1 expression in invasive breast cancer. Correlations and prognostic value. Am J Pathol 1997; 150(2):705-11. [ Google Scholar]
- Zukerberg LR, Yang WI, Gadd M, Thor AD, Koerner FC, Schmidt EV. Cyclin D1 (PRAD1) protein expression in breast cancer: approximately one-third of infiltrating mammary carcinomas show overexpression of the cyclin D1 oncogene. Mod Pathol 1995; 8(5):560-7. [ Google Scholar]
- Gillett C, Smith P, Gregory W, Richards M, Millis R, Peters G. Cyclin D1 and prognosis in human breast cancer. Int J Cancer 1996; 69(2):92-9. doi: 10.1002/(sici)1097-0215(19960422)69:2<92::aid-ijc4>3.0.co;2-q [Crossref] [ Google Scholar]
- Michalides R, Hageman P, van Tinteren H, Houben L, Wientjens E, Klompmaker R. A clinicopathological study on overexpression of cyclin D1 and of p53 in a series of 248 patients with operable breast cancer. Br J Cancer 1996; 73(6):728-34. doi: 10.1038/bjc.1996.128 [Crossref] [ Google Scholar]
- Reed W, Flørems VA, Holm R, Hannisdal E, Nesland JM. Elevated levels of p27, p21 and cyclin D1 correlate with positive oestrogen and progesterone receptor status in node-negative breast carcinoma patients. Virchows Arch 1999; 435(2):116-24. doi: 10.1007/s004280050408 [Crossref] [ Google Scholar]
- Elsheikh S, Green AR, Aleskandarany MA, Grainge M, Paish CE, Lambros MB. CCND1 amplification and cyclin D1 expression in breast cancer and their relation with proteomic subgroups and patient outcome. Breast Cancer Res Treat 2008; 109(2):325-35. doi: 10.1007/s10549-007-9659-8 [Crossref] [ Google Scholar]
- Li Z, Cui J, Yu Q, Wu X, Pan A, Li L. Evaluation of CCND1 amplification and cyclin D1 expression: diffuse and strong staining of cyclin D1 could have same predictive roles as CCND1 amplification in ER positive breast cancers. Am J Transl Res 2016; 8(1):142-53. [ Google Scholar]
- Parvin T, Das C, Choudhury M, Chattopadhyay BK, Mukhopadhyay M. Prognostic utility of cyclin D1 in invasive breast carcinoma. Indian J Surg Oncol 2019; 10(1):167-73. doi: 10.1007/s13193-018-0839-2 [Crossref] [ Google Scholar]
- Mylona E, Tzelepis K, Theohari I, Giannopoulou I, Papadimitriou C, Nakopoulou L. Cyclin D1 in invasive breast carcinoma: favourable prognostic significance in unselected patients and within subgroups with an aggressive phenotype. Histopathology 2013; 62(3):472-80. doi: 10.1111/his.12013 [Crossref] [ Google Scholar]
- Said TK, Conneely OM, Medina D, O’Malley BW, Lydon JP. Progesterone, in addition to estrogen, induces cyclin D1 expression in the murine mammary epithelial cell, in vivo. Endocrinology 1997; 138(9):3933-9. doi: 10.1210/endo.138.9.5436 [Crossref] [ Google Scholar]
- Yang C, Chen L, Li C, Lynch MC, Brisken C, Schmidt EV. Cyclin D1 enhances the response to estrogen and progesterone by regulating progesterone receptor expression. Mol Cell Biol 2010; 30(12):3111-25. doi: 10.1128/mcb.01398-09 [Crossref] [ Google Scholar]
- Roy PG, Thompson AM. Cyclin D1 and breast cancer. Breast 2006; 15(6):718-27. doi: 10.1016/j.breast.2006.02.005 [Crossref] [ Google Scholar]
- Kenny FS, Hui R, Musgrove EA, Gee JM, Blamey RW, Nicholson RI. Overexpression of cyclin D1 messenger RNA predicts for poor prognosis in estrogen receptor-positive breast cancer. Clin Cancer Res 1999; 5(8):2069-76. [ Google Scholar]
- Stendahl M, Kronblad A, Rydén L, Emdin S, Bengtsson NO, Landberg G. Cyclin D1 overexpression is a negative predictive factor for tamoxifen response in postmenopausal breast cancer patients. Br J Cancer 2004; 90(10):1942-8. doi: 10.1038/sj.bjc.6601831 [Crossref] [ Google Scholar]
- Lee A, Park WC, Yim HW, Lee MA, Park G, Lee KY. Expression of c-erbB2, cyclin D1 and estrogen receptor and their clinical implications in the invasive ductal carcinoma of the breast. Jpn J Clin Oncol 2007; 37(9):708-14. doi: 10.1093/jjco/hym082 [Crossref] [ Google Scholar]
- Yu Q, Geng Y, Sicinski P. Specific protection against breast cancers by cyclin D1 ablation. Nature 2001; 411(6841):1017-21. doi: 10.1038/35082500 [Crossref] [ Google Scholar]
- Umekita Y, Ohi Y, Sagara Y, Yoshida H. Overexpression of cyclinD1 predicts for poor prognosis in estrogen receptor-negative breast cancer patients. Int J Cancer 2002; 98(3):415-8. doi: 10.1002/ijc.10151 [Crossref] [ Google Scholar]
- Allred DC, Harvey JM, Berardo M, Clark GM. Prognostic and predictive factors in breast cancer by immunohistochemical analysis. Mod Pathol 1998; 11(2):155-68. [ Google Scholar]
- Ahlin C, Lundgren C, Embretsén-Varro E, Jirström K, Blomqvist C, Fjällskog M. High expression of cyclin D1 is associated to high proliferation rate and increased risk of mortality in women with ER-positive but not in ER-negative breast cancers. Breast Cancer Res Treat 2017; 164(3):667-78. doi: 10.1007/s10549-017-4294-5 [Crossref] [ Google Scholar]
- Bouzidi L, Triki H, Charfi S, Kridis WB, Derbel M, Ayadi L. Prognostic Value of Natural Killer Cells Besides Tumor-Infiltrating Lymphocytes in Breast Cancer Tissues. Clin Breast Cancer 2021; 21(6):e738-e47. doi: 10.1016/j.clbc.2021.02.003 [Crossref] [ Google Scholar]
- Alsaleem MA, Ball G, Toss MS, Raafat S, Aleskandarany M, Joseph C. A novel prognostic two-gene signature for triple negative breast cancer. Mod Pathol 2020; 33(11):2208-20. doi: 10.1038/s41379-020-0563-7 [Crossref] [ Google Scholar]