Arch Iran Med. 25(4):209-213.
doi: 10.34172/aim.2022.36
Original Article
Comparison of Vitamin D Levels in Premature Infants with and without Retinopathy of Prematurity
Hassan Boskabadi 1  , Majid Abrishami 2, Nasser shoeibi 2, Zahra sanei 3, Ali Moradi 4, 5, *, Maryam Zakerihamidi 6, *
, Majid Abrishami 2, Nasser shoeibi 2, Zahra sanei 3, Ali Moradi 4, 5, *, Maryam Zakerihamidi 6, * 
Author information:
1Department of Pediatrics, Faculty of Medicine, Mashhad University of Medical Sciences, Mashhad, IR Iran
2Eye Research Center, School of Medicine, Mashhad University of Medical Sciences, Mashhad, IR Iran
3School of Medicine, Mashhad University of Medical Sciences, Mashhad, IR Iran
4Orthopedic Research Center, Ghaem Hospital, Mashhad University of Medical Sciences, Mashhad, Iran
5Clinical Research Development Unit, Ghaem Hospital, Mashhad University of Medical Sciences, Mashhad, Iran
6Department of Midwifery, School of Medical Sciences, Islamic Azad University, Tonekabon, Iran
*
Corresponding Authors: Ali Moradi, MD; Orthopedic Research Center, Ghaem Hospital, Mashhad University of Medical Sciences, Mashhad, Iran/Clinical Research Development Unit, Ghaem Hospital, Mashhad University of Medical Sciences, Mashhad, Iran. Email:
ralimoradi@gmail.com; Maryam Zakerihamidi, PhD; Department of Midwifery, School of Medical Sciences, Tonekabon Branch, Islamic Azad University, Tonekabon, Iran. Cell phone: 009809113934386; Emal:
maryamzakerihamidi@yahoo.co.nz
Abstract
Background:
Retinopathy of prematurity (RoP) is a cause of newborn blindness. Several predisposing factors have been reported to contribute to the disease process. The current study aimed to compare serum vitamin D levels in infants with and without RoP.
Methods:
This case-control study was conducted on 154 very low birth weight (VLBW) infants admitted to Ghaem hospital, Mashhad, Iran, during 2016–2019. Retinal examination for RoP was done at the 32nd week of pregnancy and vitamin D level was determined using the infants’ first-day serum samples. A researcher-made questionnaire including maternal, infant, laboratory, and retinal examination information was used as the data collection tool.
Results:
Out of 154 infants in the study, 56 (36.4%) were normal while 98 (63.6%) had RoP. Based on the severity of retinopathy, 43 infants (43.9%) were at stage I, 48 (49%) at stage II, and 7 (7.1%) at stage III. Significant differences in neonatal (P<0.001) and maternal (P=0.015) vitamin D levels, first and fifth minute Apgar scores (P=0.034 and P=0.001, respectively), and weight (P=0.014) were found between the infants with and without RoP.
Conclusion:
The incidence of RoP was higher in infants with lower gestational age, lower birth weight, low first and fifth minutes Apgar scores, and male sex. Low serum levels of vitamin D in premature infants and their mothers were associated with incidence of RoP. The higher the stage of RoP, the greater was the severity of vitamin D deficiency. Thus, controlling the maternal vitamin D level during pregnancy, consumption of vitamin D supplements, and investigation of serum vitamin D levels in premature infants are recommended. Early correction of vitamin D deficiency may lead to reduction of RoP.
Keywords: Birth weight, Gestational age, Retinopathy of prematurity, Vitamin D
Copyright and License Information
© 2022 The Author(s).
This is an open-access article distributed under the terms of the Creative Commons Attribution License (
https://creativecommons.org/licenses/by/4.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Cite this article as: Boskabadi H, Abrishami M, Shoeibi N, Sanei Z, Moradi A, Zakerihamidi M. Comparison of vitamin d levels in premature infants with and without retinopathy of prematurity. Arch Iran Med. 2022;25(4):209-213. doi: 10.34172/aim.2022.36
Introduction
With the advancing knowledge of perinatologyin recent decades, maintaining the survival of high-risk neonates including premature infants has been made possible.1 Very low birth weight (VLBW) infants have many more problems and require greater care.2 Retinopathy of Prematurity (RoP).3 is one of the serious problems among VLBW infants and a leading cause of childhood blindness. RoP, as a side effect of oxygen therapy, can lead to blindness in the incompletely vascularized retina in premature infants.4
The incidence and severity of RoP increases by reduction in gestational age and birth weight. In a previous study in Iran, the prevalence of RoP in infants under 32 weeks was reported at about 44%.2 Although many factors are involved in the incidence of RoP, low gestational age, LBW, and oxygen use are important factors in developing RoP. Other possible important factors include: apnea, mechanical ventilation, anemia and need for blood transfusion, vitamin deficiencies, intraventricularhemorrhage, sepsis, acidosis, hypotension, pneumothorax, chronic lung diseases, high levels of arterial carbon dioxide, seizure, and bradycardia.5,6 Effective early identification of high risk infants and examination of their retina has been proven to reduce the complications of RoP.7,8
Maintaining normal blood levels of calcium and phosphorus is the main biological function of vitamin D. Vitamin D promotes an overall boost in the immune system. Considering the numerous biologic functions of vitamin D in body metabolism, the need for it increases during pregnancy. Moreover, the fetus is unable to produce 25-hydroxy vitamin D and relies completely on its maternal transfer.9 Also, as placental transfer of vitamin D mostly occurs during the third trimester, vitamin D deficiency is one of the problems of premature infants.10 Maternal vitamin D deficiency leads to reduction in placental vitamin D transfer.11
The risk factors for maternal and consequently neonatal vitamin D deficiency include low exposure to sunlight, regular sunscreen use, residence in northern latitudes, dark skin, obesity, multilayer clothing, aging, malnutrition, malabsorption syndrome, and medications.12 In neonates, vitamin D deficiency accompanies intrauterine growth restriction, weight loss, and increased risk of preterm labor, leading to preterm labor, respiratory distress syndrome, and increased bronchopulmonary dysplasia.13 Low serum levels of 25-hydroxy vitamin D on the first day of life have been proposed as a potential risk factor for retinopathy of immaturity, and hence, treatment of immature neonates is necessary.14 Expression of vitamin D receptor has been shown to play an important role in the evolution of retinal vessels, especially during the final steps of retinal vascularization. Although pathologic retinal neovascularization during oxygen-therapy induced ischemic retinopathy is independent of the expression of vitamin D receptor, the inhibition of retinal neovascularization through 1,25 hydroxy vitamin D3 depends on the expression of vitamin D receptor.15
Considering the placental passage of the highest levels of calcium and vitamin D during the third trimester, premature infants are most likely to have problems in this regard. On the other hand, these problems would be exacerbated in cases of maternal vitamin D deficiency. As vitamin D deficiency can be a potential risk factor for RoP, the current case-control study attempted to evaluate the vitamin D levels in infants with and without RoP.
Materials and Methods
This study was conducted on VLBW infants admitted to the intensive care unit at Ghaem hospital, Mashhad, Iran, during 2016–2019.
Due to the lack of a similar study and according to Kabataş et al14 who reported the vitamin D level in infants with RoP who needed or did not need treatment at 7.1 ± 5.2 ng/dL and 11.9 ± 6.5 ng/dL, respectively, using the student t test with α = 0.01 and β = 0.1, the sample size in each group was calculated as 46 infants. However, considering the probable dropout, the final sample size was adjusted to 56 infants in each group.
Prepartum maternal whole blood samples (2 mL) were collected. Serum samples were also taken from infants < 1500 g for vitamin D measurement along with sampling for other routine tests in the first hour of life. The serum samples were stored at -20°C for further laboratory evaluation. Vitamin D measurement was performed by ELISA (RT2100C, Germany). The VLBW infants were allocated into four groups according to their vitamin D levels: severe deficiency (≤ 10 ng/mL), moderate deficiency (10.1–20 ng/mL), mild deficiency (20.1–30 ng/mL) and normal (> 30 ng/mL).
The demographic data of infants were recorded in a checklist. The characteristics of infants (birth weight, age, gender, gestational age, Apgar score, and clinical symptoms), maternal history (age, pregnancy and delivery problems, mode of delivery), and laboratory results were collected and recorded in a questionnaire. The infant’s condition was monitored up to discharge time. Screening for RoP was started between 4–6 weeks after birth. A single drop of cyclopentolate hydrochloride/phenylephrine (2 mg.mL -1/2.5 mg.mL -1) was distilled into each eye 30 minutes before the examination to induce mydriasis. Fundal examination using an indirect ophthalmoscope and a 20D lens was performed by a single investigator (MA) at Khatam-al-Anbia hospital, a tertiary referral eye hospital. The examinations continued until full vascularization of the retina or the post-conception age of 40 weeks according to an accurate gestational age. Cases with RoP were treated according to the severity and diagnostic criteria. The infant was allocated in the control group if the primary retinal examination was normal. RoP was defined based on the international classification.16
The severity of RoP was defined in five stages through which the disease serially passes. However, stages I and IV may be present simultaneously in one eye17:
-
Stage I (presence of a demarcation line)
-
Stage II (presence of ridge ± fibrous - vesicular proliferative small tissues)
-
Stage III (ridge with extra-retinal fibro-vesicular proliferation)
-
Stage IV (retinal sub-total detachment A, without fovea involvement & B with foveal involvement)
-
Stage V (total retinal detachment).
Plus disease is the severity index of the disease and is defined as intravascular dilation and arteriolar tortuosity of the posterior vessels.18
Following data collection and recording in SPSS 21, normality (Kolmogorov-Smirnov) test, Pearson’s correlation coefficient, and independent t test were used for analysis of the relationship between the variables. The Kolmogorov-Smirnov test was used to assess the normality of data distribution. In case of non-normal distribution, non-parametric tests including Spearman’s correlation coefficient and Mann-Whitney tests were used.
The association of the delivery and gender with RoP condition was assessed using the chi-square or Fisher’s exact tests. After checking the homogeneity of variances (Levene’s test), analysis of co-variances was used to control the gestational age, first and fifth minute Apgar scores, and duration of O2 therapy. P < 0.05 was considered as the minimum significant level in this study.
Results
Among 154 participants in the study, 56 infants (36.4%) were normal and 98 infants (63.6%) had RoP. Based on the severity of retinopathy, 43 infants (43.9%) were stage I, 48 infants (49%) were stage II, and 7 infants (7.1%) were stage III. Plus disease was present in five cases (5.1%). In terms of zone of RoP, 3.8% was zone 1, 46.3% zone 2, 45% zone 3, 1.3% zone 1–2, and 3.8% was zone 2–3. In the follow-up of infants with RoP, 91 infants improved, five infants needed bevacizumab (Avastin) injection, and two patients had surgery. In terms of RoP prognosis, 96.5% were normal and 3.5% progressed to blindness.
Statistically significant differences were found between the infants with and without RoP in terms of neonatal vitamin D (P < 0.001, CI: 7.96–13.79), maternal vitamin D (P = 0.015), gestational age (P < 0.001, CI: 2.12–18.73), first minute Apgar score (P = 0.034, CI: 0.05–1.37), fifth minute Apgar score (P < 0.001, CI: 0.29–1.31), and birth weight (P = 0.014, CI: 35.75–279.0) (Table 1). Analysis of co-variances with the elimination of factors affecting the gestational age, first and fifth minute Apgar scores, and duration of O2 therapy showed a significant difference in the serum vitamin D level (P = 0.013) between the RoP and normal neonates.
Table 1.
Comparison of Average Neonatal and Maternal Variables between Groups of Infants with and without RoP
|
Variables
|
Infants without RoP
56 (36.4%)
|
Infants with RoP
98 (63.6%)
|
Mean Differences (95% CI)
|
P
Value
a
|
| Neonatal vitamin D (ng/dL) |
22.33 ± 11.40 |
11.46 ± 6.50 |
10.87 (7.96–13.79) |
0.000 |
| Maternal vitamin D (ng/dL) |
29.66 ± 14.49 |
19.23 ± 14.47 |
10.43 (2.12–18.73) |
0.015 |
| Gestational age (wk) |
32.05 ± 1.90 |
30.58 ± 2.20 |
1.47 (0.76–2.17) |
0.000 |
| First minute Apgar score |
6.87 ± 1.81 |
6.15 ± 2.12 |
0.72 (0.05–1.37) |
0.034 |
| Fifth minute Apgar score |
8.36 ± 1.11 |
7.55 ± 1.66 |
0.81 (0.29–1.31) |
0.001 |
| Birth weight |
1491.87 ± 382.80 |
1334.58 ± 353.85 |
157.29 (35.75–279.0) |
0.014 |
RoP, Retinopathy of prematurity.
Values are in terms of mean ± SD; at test.
About 59% of infants with RoP and 63% of those without RoP were born through cesarean section. In addition, 51% of infants with RoP and 43% of those without RoP were male. Although cesarean delivery (P = 0.769) and male gender (P = 0.352) were more prevalent among infants with RoP, the difference between the two groups was not statistically significant. According to Table 2, the higher the RoP grade, the lower was the vitamin D level.
Table 2.
Average Vitamin D Levels at Different Retinopathy of Prematurity Severities
|
Global Exam
|
Mean±SD
|
| Normal |
22.33 ± 11.40 |
| RoP 1 |
14.06 ± 7.79 |
| RoP 2 |
11.43 ± 7.49 |
| RoP 3 |
8.20 ± 0.14 |
ROP, retinopathy of prematurity.
Vitamin D deficiency showed a significant association with the incidence of RoP (Pearson’s correlation = -0.518, P < 0.001). Almost 83% of infants with severe Vit D deficiency suffered from RoP while 17.2% had normal retina; however, only 11.1% of infants with normal vitamin D levels had RoP and 88.9% had normal retina (Figure 1). The prevalence of strabismus increased by 20% in LBW infants, and this increase was related to the RoP stage, as the incidence of strabismus was 6% in stage I and 30% in stage III RoP.
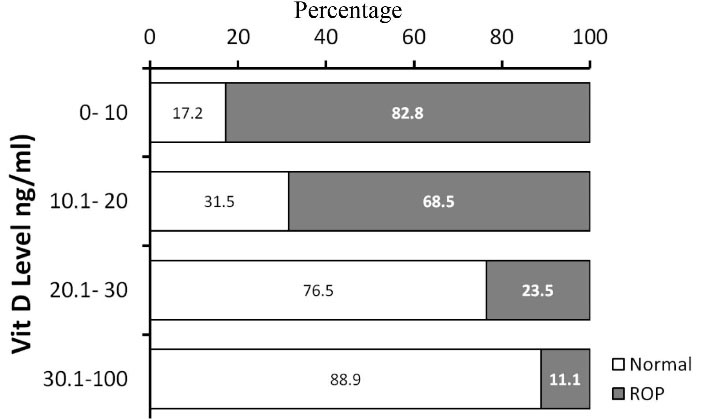
Figure 1.
Distribution of RoP among Infants with Different Levels of Vitamin D.
.
Distribution of RoP among Infants with Different Levels of Vitamin D.
Discussion
This study aimed to assess the relationship between neonatal serum vitamin D levels and RoP. According to our findings, infants with RoP had significantly lower serum vitamin D levels compared to those without RoP. Also, vitamin D level was strongly associated with the incidence of RoP (Pearson correlation: 0.518). The severity of RoP increased with reduction in serum vitamin D. Therefore, vitamin D deficiency can be suggested as a risk factor for prediction of neonatal RoP.
A significant relationship was shown between the incidence of RoP and infant’s weight. The prevalence of RoP was higher with lower birth weight. This result is consistent with the findings of several previous studies.19-22
Forty-four percent of our newborns had RoP stage I, 49% stage II, and 7.1% stage III. The prevalence of RoP in a previous study was 68% in stage I, 23.5% in stage II, and 8.5% in stage III.23 Also, another study reported that 30.2% of infants under 1500 g suffered from stage I and II RoP and 16.6% of them suffered from stage III and above. In infants weighing 1500 to 2500 g, 3.2% had stage I and II RoP, and 0.6% had stage III and above.24
Consistent with the findings of a previous report, the incidence of RoP in our study was higher at lower gestational ages.19 Low gestational age, LBW, and receiving oxygen therapy are risk factors for the incidence of RoP.25 The incidence of RoP has been reported at 44% in infants with gestational age lower than 30–32 weeks or a birth-weight less than 1500 g.26,27
In the follow-up phase, 92.86% of infants with RoP improved, 5.1% needed Avastin injections, and 2.04% had surgery. Fortunately, RoP improved without visual impairment in most cases (80%)3 The prevalence of strabismus increased in LBW infants by 20%. This increase was related with the RoP stage, as the incidence of strabismus in stage I RoP was 6%, while, it increased to 30% in stage III. However, early treatment has been shown to improve the vision even in the eyes with poor visual function.28 In a previous study with 70% RoP in premature infants, 68% recovered and 32% needed treatment for RoP (75% of infants were treated by Avastin injection and 25% by laser).29
Consistent with other studies, our findings showed no significant relationship between gender and RoP incidence.30,31
Serum levels of vitamin D in infants with RoP were lower in our study regardless of gestational age. The severity of vitamin D deficiency increased with higher stages of RoP. To the best of our knowledge, no relationship between vitamin D deficiency and RoP has been reported before. As vitamin D directly affects the vascular endothelial stability, its deficiency will most likely affect the incidence of RoP in premature infants who are at risk of vitamin D deficiency.32 The inflammatory and angiogenic effects of vitamin D deficiency can cause early damage to the retinal blood vessels.33 In vitro studies have shown that vitamin D improves the angiogenic properties of precursor cells through upregulation of the expression of vascular endothelial growth factor. Since vitamin D deficiency is associated with degeneration of endothelial function, the number of endothelial progenitor cells can also be reduced.34 The possible mechanisms by which vitamin D can cause small vessels damage include inflammation, fat metabolism, and the renin-angiotensin-aldosterone system.35
As a limitation, the present study failed to consider the gestational age and birth weight of the participant neonates. Future studies with subject matching in terms of gestational age and birth weight of neonates are recommended.
In conclusion, the incidence of RoP was higher in infants with lower gestational age, lower birth weight, low first and fifth minutes Apgar scores, and male sex. Low serum levels of vitamin D in premature infants and their mothers were associated with incidence of RoP. The higher the stage of RoP, the greater was the severity of vitamin D deficiency. Thus, controlling the maternal vitamin D level during pregnancy, consumption of vitamin D supplements, and investigation of serum vitamin D levels in premature infants are recommended. Early correction of vitamin D deficiency may lead to reduction of RoP.
Acknowledgements
The authors would like to appreciate the support from the Clinical Research Development Unit, Ghaem Hospital, Mashhad University of Medical Sciences, Mashhad, Iran.
Authors’ Contribution
HB, MA, NS, ZS, AM, MZ conceptualized and designed the study, drafted the initial manuscript, and approved the final manuscript as submitted. HB, ZS, AM, and MZ designed the study and carried out the initial analyses, reviewed and revised the manuscript, and approved the final manuscript as submitted. HB, MA and NS carried out the initial analyses, reviewed and revised the manuscript, and approved the final manuscript as submitted. HB, MA, NS, ZS, AM and MZ designed the data collection instruments, and coordinated and supervised data collection at two of the four sites, critically reviewed the manuscript, and approved the final manuscript as submitted. All authors approved the final manuscript as submitted and agree to be accountable for all aspects of the work.
Conflict of Interest Disclosures
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Statement
The study was approved by the ethics committee of Mashhad University of Medical Sciences (IR.MUMS.fm.REC.1395.631). All parents were provided a written informed consent to sign.
References
- Boskabadi H, Bagheri F, Askari Hosseni Z. Developmental disorders in preterm neonates during the first two years of life using the ages and stages questionnaire. J Babol Univ Med Sci 2016; 18(2):7-13. doi: 10.22088/jbums.18.2.7 [Crossref] [ Google Scholar]
- Abrishami M, Maemori GA, Boskabadi H, Yaeghobi Z, Mafi-Nejad S, Abrishami M. Incidence and risk factors of retinopathy of prematurity in Mashhad, Northeast Iran. Iran Red Crescent Med J 2013; 15(3):229-33. doi: 10.5812/ircmj.4513 [Crossref] [ Google Scholar]
- Multicenter Study Group. Supplemental Therapeutic Oxygen for Prethreshold Retinopathy of Prematurity (STOP-ROP), a randomized, controlled trial. I: primary outcomes. Pediatrics 2000;105(2):295-310. 10.1542/peds.105.2.295
- Phelps DL. Retinopathy of prematurity: history, classification, and pathophysiology. NeoReviews 2001; 2(7):e153-e66. doi: 10.1542/neo.2-7-e153 [Crossref] [ Google Scholar]
- Boskabadi H, Pourbadakhshan N, Pishghadam E, Zakerihamidi M. Comparing nucleated red blood cells in neonates with and without retinopathy. Curr Pediatr Rev. 2021. 10.2174/1573396317666210909155024
- Boskabadi H, Marefat M, Maamouri G, Abrishami M, Abrishami M, Shoeibi N. Evaluation of pro-oxidant antioxidant balance in retinopathy of prematurity. Eye (Lond) 2022; 36(1):148-52. doi: 10.1038/s41433-021-01465-6 [Crossref] [ Google Scholar]
- Simon J. Basic and Clinical Science Course, 2004-2005: Pediatric Ophthalmology and Strabismus Section 6. San Francisco, CA: American Academy of Ophthalmology; 2004. p.307-14.
- Thomas J, Gregory L, Louis B. 2007-2008 Basic and Clinical Science Course Section 13: Refractive Surgery. San Francisco, CA: American Academy of Ophthalmology; 2007.
- Weisman Y. Maternal, fetal and neonatal vitamin D and calcium metabolism during pregnancy and lactation. Endocr Dev 2003; 6:34-49. doi: 10.1159/000072768 [Crossref] [ Google Scholar]
- Kaushal M, Magon N. Vitamin D in pregnancy: a metabolic outlook. Indian J Endocrinol Metab 2013; 17(1):76-82. doi: 10.4103/2230-8210.107862 [Crossref] [ Google Scholar]
- Onwuneme C, Martin F, McCarthy R, Carroll A, Segurado R, Murphy J, et al. The association of vitamin D status with acute respiratory morbidity in preterm infants. J Pediatr 2015;166(5):1175-80.e1. 10.1016/j.jpeds.2015.01.055
- Mulligan ML, Felton SK, Riek AE, Bernal-Mizrachi C. Implications of vitamin D deficiency in pregnancy and lactation. Am J Obstet Gynecol 2010;202(5):429.e1-429.e9. 10.1016/j.ajog.2009.09.002
- Lykkedegn S, Sorensen GL, Beck-Nielsen SS, Christesen HT. The impact of vitamin D on fetal and neonatal lung maturation. A systematic review. Am J Physiol Lung Cell Mol Physiol 2015; 308(7):L587-602. doi: 10.1152/ajplung.00117.2014 [Crossref] [ Google Scholar]
- Kabataş EU, Dinlen NF, Zenciroğlu A, Dilli D, Beken S, Okumuş N. Relationship between serum 25-hydroxy vitamin D levels and retinopathy of prematurity. Scott Med J 2017; 62(4):129-35. doi: 10.1177/0036933017701867 [Crossref] [ Google Scholar]
- Jamali N, Wang S, Darjatmoko SR, Sorenson CM, Sheibani N. Vitamin D receptor expression is essential during retinal vascular development and attenuation of neovascularization by 1, 25(OH)2D3. PLoS One 2017; 12(12):e0190131. doi: 10.1371/journal.pone.0190131 [Crossref] [ Google Scholar]
- Aaberg T, Ben-Sira I, Charles S, Clarkson J, Cohen BZ, Flynn J. An international classification of retinopathy of prematurity: II. The classification of retinal detachment. Arch Ophthalmol 1987; 105(7):906-12. doi: 10.1001/archopht.1987.01060070042025 [Crossref] [ Google Scholar]
- Riordan-Eva P, Whitcher JP. Vaughan & Asbury’s General Ophthalmology. Wiley Online Library; 2008.
- Shah PK, Prabhu V, Karandikar SS, Ranjan R, Narendran V, Kalpana N. Retinopathy of prematurity: past, present and future. World J Clin Pediatr 2016; 5(1):35-46. doi: 10.5409/wjcp.v5.i1.35 [Crossref] [ Google Scholar]
- Naderian G, Iranpour R, Mohammadizadeh M, Najafabadi FF, Badiei Z, Naseri F, et al. The frequency of retinopathy of prematurity in premature infants referred to an ophthalmology clinic in Isfahan. J Isfahan Med Sch 2011;29(128). 10.29252/ajcm.26.1.44
- Holmström G, el Azazi M, Jacobson L, Lennerstrand G. A population based, prospective study of the development of ROP in prematurely born children in the Stockholm area of Sweden. Br J Ophthalmol 1993; 77(7):417-23. doi: 10.1136/bjo.77.7.417 [Crossref] [ Google Scholar]
- O’Connor MT, Vohr BR, Tucker R, Cashore W. Is retinopathy of prematurity increasing among infants less than 1250 g birth weight?. J Perinatol 2003; 23(8):673-8. doi: 10.1038/sj.jp.7211008 [Crossref] [ Google Scholar]
- Mousavi Z, Karkhaneh R, Riazi-Esfahani M, Mansouri M, Roohipoor R, Ghalichi L, et al. Incidence, severity and risk factors for retinopathy of prematurity in premature infants with late retinal examination. Bina J Ophthalmol 2008;13(4):412-7. [Persian].
- Conrath JG, Hadjadj EJ, Forzano O, Denis D, Millet V, Lacroze V. Screening for retinopathy of prematurity: results of a retrospective 3-year study of 502 infants. J Pediatr Ophthalmol Strabismus 2004; 41(1):31-4. doi: 10.3928/0191-3913-20040101-08 [Crossref] [ Google Scholar]
- Andruscavage L, Weissgold DJ. Screening for retinopathy of prematurity. Br J Ophthalmol 2002; 86(10):1127-30. doi: 10.1136/bjo.86.10.1127 [Crossref] [ Google Scholar]
- Fouladinejad M, Motahari M, Gharib M, Sheishari F, Soltani M. The prevalence, intensity and some risk factors of retinopathy of premature newborns in Taleghani hospital, Gorgan, Iran. J Gorgan Univ Med Sci 2009;11(2):51-4. [Persian].
- Gonçalves E, Násser LS, Martelli DR, Alkmim IR, Mourão TV, Caldeira AP. Incidence and risk factors for retinopathy of prematurity in a Brazilian reference service. Sao Paulo Med J 2014; 132(2):85-91. doi: 10.1590/1516-3180.2014.1322544 [Crossref] [ Google Scholar]
- Maini B, Chellani H, Arya S, Guliani BP. Retinopathy of prematurity: risk factors and role of antenatal betamethasone in Indian preterm newborn babies. J Clin Neonatol 2014; 3(1):20-4. doi: 10.4103/2249-4847.128724 [Crossref] [ Google Scholar]
- Heidary F, Gharebaghi R. Outcomes of retinopathy of prematurity. Med Hypothesis Discov Innov Ophthalmol 2016; 5(4):112-4. [ Google Scholar]
- Ahmadpour-Kacho M, Zahed Pasha Y, Rasoulinejad SA, Hajiahmadi M, Pourdad P. Correlation between retinopathy of prematurity and clinical risk index for babies score. Tehran Univ Med J 2014;72(6):404-11. [Persian].
- Hakeem AH, Mohamed GB, Othman MF. Retinopathy of prematurity: a study of prevalence and risk factors. Middle East Afr J Ophthalmol 2012; 19(3):289-94. doi: 10.4103/0974-9233.97927 [Crossref] [ Google Scholar]
- Daraie G, Nooripoor S, Ashrafi AM, Ghorbani R. Incidence of retinopathy of prematurity and some related factors in premature infants born at Amir-al-Momenin hospital in Semnan, Iran. Koomesh journal 2016;17(2):297-303. [Persian].
- Gibson CC, Davis CT, Zhu W, Bowman-Kirigin JA, Walker AE, Tai Z. Dietary Vitamin D and Its Metabolites Non-Genomically Stabilize the Endothelium. PLoS One 2015; 10(10):e0140370. doi: 10.1371/journal.pone.0140370 [Crossref] [ Google Scholar]
- Kaur H, Donaghue KC, Chan AK, Benitez-Aguirre P, Hing S, Lloyd M. Vitamin D deficiency is associated with retinopathy in children and adolescents with type 1 diabetes. Diabetes Care 2011; 34(6):1400-2. doi: 10.2337/dc11-0103 [Crossref] [ Google Scholar]
- Dusso AS. Renal vitamin D receptor expression and vitamin D renoprotection. Kidney Int 2012; 81(10):937-9. doi: 10.1038/ki.2012.30 [Crossref] [ Google Scholar]
- Kassi E, Adamopoulos C, Basdra EK, Papavassiliou AG. Role of vitamin D in atherosclerosis. Circulation 2013; 128(23):2517-31. doi: 10.1161/circulationaha.113.002654 [Crossref] [ Google Scholar]