Arch Iran Med. 25(3):155-160.
doi: 10.34172/aim.2022.26
Original Article
Effects of Renal Cysts on Renal Function
Miaomiao Zhu 1, Xu Chu 1, Chen Liu 1, * 
Author information:
1Department of Abdominal Ultrasound, The Affiliated Hospital of Qingdao University, Qingdao University, Qingdao, China
*Corresponding Author: Chen Liu, MD; Department of Abdominal Ultrasound, The Affiliated Hospital of Qingdao University, Qingdao University, Qingdao 266000, China. Tel:+86-15811176878; Email:
guanjunhou@qdu.edu.cn
Abstract
Background:
People with simple renal cysts (SRCs) are more likely to develop renal injury and dysfunction, but the mechanisms remain controversial, as the process of SRC formation and its characteristics are not yet well-known. This research focuses on the characteristics of SRCs and discusses how SRCs cause renal injury.
Methods:
In 2014, a total of 401 participants without any prior kidney disease were selected for the research. Their average age was 49.7 years. SRC morphology and changes were monitored over a 5-year follow-up period. Renal volume and blood perfusion were measured by ultrasound imaging. Logistic regression analysis was used to assess the relationship between renal cyst and renal function.
Results:
During the 5-year follow-up, elderly participants with multiple cysts (odds ratio [OR] 1.89; 95% CI 1.67 to 5.99) and a maximum cyst diameter of 1.5 cm or greater (OR 1.93; 95% CI 1.15 to 5.34) were found to be positively correlated with renal injury. A decrease in intrarenal perfusion was observed at the early stages of follow-up, but the reduction in renal volume was a slow and gradual process.
Conclusion:
Elderly people with multiple cysts, a maximum cyst diameter of the cysts≥1.5 cm, and multiple cysts are more likely to suffer renal injury. Ultrasound examination has an important status in monitoring the changes in renal volume and peak systolic velocity (PSV) of the renal interlobar artery.
Keywords: Renal injury, Simple renal cyst, Ultrasonography
Copyright and License Information
© 2022 The Author(s).
This is an open-access article distributed under the terms of the Creative Commons Attribution License (
https://creativecommons.org/licenses/by/4.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Cite this article as: Zhu M, Chu X, Liu C. Effects of renal cysts on renal function. Arch Iran Med. 2022;25(3):155-160. doi: 10.34172/aim.2022.26
Introduction
According to the Bosniak classification system, simple renal cysts (SRCs) are divided into stage I and stage II types. They are usually well-shaped round cysts with smooth cyst walls and no deposit sediment inside.1 SRCs account for about 70% of benign neoplastic lesions of the kidney, which can present at all ages. With advancing age, SRCs are often inadvertently detected in the process of routine health examinations. However, the specific mechanisms of cyst formation are unclear. Some cysts tend to increase, causing symptoms such as pain, hematuria, and/or urinary obstruction.2,3 Furthermore, previous studies have shown that SRCs are also an important cause of renal dysfunction.4 Other recent evidence suggests that SRCs can cause other kidney-related symptoms, such as hypertension, and changes in kidney volume and blood perfusion, which in turn affect kidney function.5-7
However, the role and possible mechanisms underlying SRC development are still controversial.8 In addition, the characteristics of renal cysts that cause renal dysfunction are still unclear. This research discusses the characteristics of renal cysts and the relationship between SRCs and renal injury.
Materials and Methods
Study Population
This was a retrospective study examining 1002 patients with renal cysts from September 2014 to September 2019. The patients under 80 years of age who had normal renal function, no history of renal disease, no medical treatment plans for the duration of this study, and whose clinical data were available through our hospital information system were enrolled into the study. Patients with proteinuria with or without reduced glomerular filtration rate, and patients with polycystic kidney, solitary kidney, or multiple organ failure were not enrolled in this study. Patients with nephrectomy, or irregularly shaped kidneys, or those whose renal measurements were not reproducible were excluded. Patients with any other major diseases were also excluded.
Finally, 401 participants were enrolled in the study (209 males and 192 females, aged 21–79 years) and followed up for five years. Patients who presented with proteinuria with or without decreased estimated glomerular filtration rate (eGFR) during follow-up were defined as experiencing renal injury.
Blood Biochemistry Measurements
Renal insufficiency was defined as eGFR less than 60 mL/min/1.73 m2. The amount of protein in the urine was classified as negative, trace, 1 + , 2 + , 3 + , or 4 + . If the urine test showed 1 + , the patients were considered to have albuminuria. Any patient suffering from proteinuria or renal insufficiency was defined as experiencing renal injury. Fasting venous blood (5 mL) was collected prior to 10:00 AM. An automatic biochemical analyzer was used to analyze fasting blood glucose. A questionnaire was used to assess the patient’s physical condition and habits. Fasting blood glucose of 7.0 mmol/L was defined as diabetes. Systolic blood pressure ≥ 140 mmHg and/or diastolic blood pressure ≥ 90 mm Hg were defined as hypertension.
Equipment
The GE Log E9 Color Doppler ultrasonic diagnostic instrument (GE Healthcare, Milwaukee, WI, USA) with a 3.5 MHz transducer was used to diagnose SRCs.
Subgroup Criteria
According to the characteristics of cysts, renal cysts were divided into different groups. The presence of more than two cysts was defined as multiple cysts. In addition, we measured the maximum diameter of the cyst.
Statistical Methods
The SPSS 19 statistical analysis software (IBM, Armonk, NY, USA) was used to analyze the data. The Kolmogorov–Smirnov test was used to assess the normality of the data, and the P values of the progressive significance (2-tailed) tests were calculated. If the P value was less than 0.05, the results were considered not in accordance with a normal distribution.
Data for non-normal distributions are expressed as medians and quartile ranges (QR). The independent sample t test was used for comparison of normal data, and the rank sum test was used for comparison of abnormal data. The chi-square test was used to test the difference of classification variable distribution.
The risk factors of kidney damage were estimated by univariate and multivariate logistic regression analyses. Related clinical studies have confirmed the relevant variables or shown in univariate analysis that the variables related to the purpose of the study are considered baseline variables, which eventually enter the multivariate logistic regression model for analysis. Considering the brevity of the final research model, we carefully selected the variables needed for the final analysis. We focused on the analysis of P value < 0.1 candidate variables in univariate logistic regression. We set the criteria for confounder variables to P values greater than 0.05 and less than 0.1. Age, sex, hypertension, diabetes, smoking, etc. should be seen as independent variables. Rank-sum tests and repeated measurement variance analyses were used to compare the changes in renal volume and the peak systolic velocity (PSV) of the renal interlobar artery between the renal injury group and the non-renal injury group.
Results
Clinical Data
Among the 401 participants, the mean age was 49.7 (5.5) years, and 52% were male. The prevalence of renal injury was 10% in the total population. The baseline characteristics of patients are shown in Table 1. The data to be included in the study were tested for normality. In the progressive significance (2-tailed) test, the P value was less than 0.05, suggesting that the results were not in accordance with normal distribution. The average age of the renal injury group was significantly higher than that of the non-renal injury group (57.7 vs. 47.5 years, P < 0.001). Additionally, the proportion of males was significant higher in the renal injury group compared to the non-renal injury group (75% vs, 49.5%, P < 0.001). The prevalence rates of hypertension, diabetes, smoking, and drinking were also higher in the renal injury group compared to the non-renal injury group (P < 0.001). The mean values of uric acid, body mass index (BMI), blood glucose and proteinuria were also significantly higher in the renal injury group than the non-renal injury group (P < 0.001). The mean eGFR of the renal injury group was lower than that of the non-renal injury group (P < 0.001).
Table 1.
The Baseline Characteristics of Patients with Renal Cysts
|
Characteristics
|
Total (n=401)
|
Non-renal injury (n=361)
|
Renal Injury (n=40)
|
P
Value
|
| Male |
209 (52%) |
179 (49.6%) |
30 (73%) |
< 0.001 |
| Age (y) |
49.7 (5.5) |
47.5 (5.9) |
57.7 (8.5) |
< 0.001 |
| BMI (kg/m2) |
25.6 (3.4) |
24.9 (1.6) |
27.1 (5.1) |
< 0.001 |
| Smoking (n %) |
211 (52.6%) |
185 (51.2%) |
26 (65%) |
< 0.001 |
| Drinking (n %) |
187 (46.6%) |
156 (43.2%) |
31 (77.5%) |
< 0.001 |
| Diabetes (n %) |
91 (22.7%) |
72 (19.9%) |
19 (47.5%) |
< 0.001 |
| Blood glucose (mmol/L) |
5.72 (1.1) |
5.51 (1.6) |
5.93 (1.3) |
< 0.001 |
| Hypertension (n %) |
82 (20.4%) |
50 (13.8%) |
32 (78.0%) |
< 0.001 |
| Uric acid (mg/dL) |
5.2 (1.3) |
5.1 (1.1) |
5.6 (1.3) |
< 0.001 |
| Baseline eGFR (mL/min/1.73 m2) |
99.2 (14.7) |
103.2 (13.3) |
95.9 (15.9) |
< 0.001 |
| Proteinuria(g/L) |
71 (9.8) |
64.9 (7.7) |
106.1 (10.6) |
< 0.001 |
| Renal cysts |
|
|
|
|
| Multiple cysts (≥ 2) |
159 |
127 (35.2%) |
32 (80%) |
< 0.001 |
| Maximum diameter (≥ 1.5 cm) |
58 |
47 (13.1%) |
11 (27.5%) |
< 0.001 |
BMI, body mass index; eGFR, estimated glomerular filtration rate
Renal Cysts and Renal Injury
Univariate logistic regression analyses were performed. Several variables, including age, sex, hypertension, and diabetes, showed P values less than 0.1 and were therefore considered as confounder factors and included in the multivariate model. The P value of renal cysts was less than 0.05, and the univariate logistic regression showed that SRCs were independently associated with renal injury with odds ratio (OR) of 2.29 and 95% confidence interval (CI) ranging from 1.33 to 2.81. Multivariable logistic regression was applied to subgroup analysis. Multiple cysts (OR 2.25; 95% CI 1.06–8.73) and cysts with a maximum diameter ≥ 1.5 cm (OR 2.11; 95%CI 1.71–5.69) were positively correlated with renal injury. After adjusting for age, sex, hypertension, and diabetes, the OR of multiple cysts, and cysts with maximum diameters of 1.5 cm or greater were 1.89 (95% CI 1.67–5.99) and 1.93 (95% CI 1.15–5.34), respectively (Table 2).
Table 2.
Multivariable Logistic Regression Analysis for the Association Between Renal Cysts and Renal Injury
|
Participants
|
Crude OR (95% CI)
|
Multivariable adjusted OR
*
|
| Multiple cysts (≥ 2) |
2.25 (1.06–8.73) |
1.89 (1.67–5.99) |
Maximum diameter
(≥ 1.5 cm) |
2.11 (1.71–5.69) |
1.93 (1.15–5.34) |
OR, odds ratio; CI, confidence interval.
*OR was adjusted for age, sex, hypertension, and diabetes.
The differences in the change of renal volume between the renal injury group and the non-renal injury group did not satisfy the spherical test (Greenhouse-Geisser = 0.763, P = 0. 001). Multivariate analysis of variance (ANOVA) showed that the differences in the data at different time points were statistically significant (P = 0.000, F = 42.72), and time was related to changes in renal function (P = 0.000, F = 2.41). The changes in renal volume during follow-up between the two groups are shown in Table 3. There was no significant difference in renal volume between the two groups before follow-up (P > 0.05).
Table 3.
Changes in the Renal Volume and the Peak Systolic Velocity of the Renal Interlobar Artery
|
Time
|
Renal Volume
|
Peak Velocity of Interlobar Artery Contraction
|
|
Renal Injury
|
Non-renal Injury
|
Renal Injury
|
Non-renal Injury
|
| 1 |
71.1 (23.1) |
70.1 (18.1) |
33.4 (6.7) |
31.6 (5.9) |
| 2 |
68.9 (20.5) |
69.9 (18.6) |
32.8 (7.1) |
32.2 (6.3) |
| 3 |
66.7 (21.6) |
69.8 (18.1) |
32.2 (8.4) |
32.9 (5.5) |
| 4 |
64.9 (18.9) |
69.9 (19.2) |
26.5 (6.9) |
32.3 (6.1) |
Changes in the renal volume and the peak systolic velocity (PSV) of the renal interlobar artery were monitored by ultrasound during follow-up.
Analysis of the trend in renal volume in the two groups revealed that the renal volume in the renal injury group decreased gradually with time, but the volume in the non-renal injury group did not change significantly (Figure 1A).
The differences in the change of the PSV of the renal interlobar artery between the renal injury group and non-renal injury group did not satisfy the spherical test (Greenhouse-Geisser = 0.911, P = 0. 000), and multivariate ANOVA was performed. The results showed that the differences in the data at different time points were statistically significant (P = 0.000, F = 20.8), and time was related to changes in the PSV of the renal interlobar artery (P = 0.000, F = 5.29). The changes in the PSV of the renal interlobar artery during follow-up between the two group are shown in Table 3. There was no significant difference in the PSV of the renal interlobar artery between the two groups before follow-up (P > 0.05).
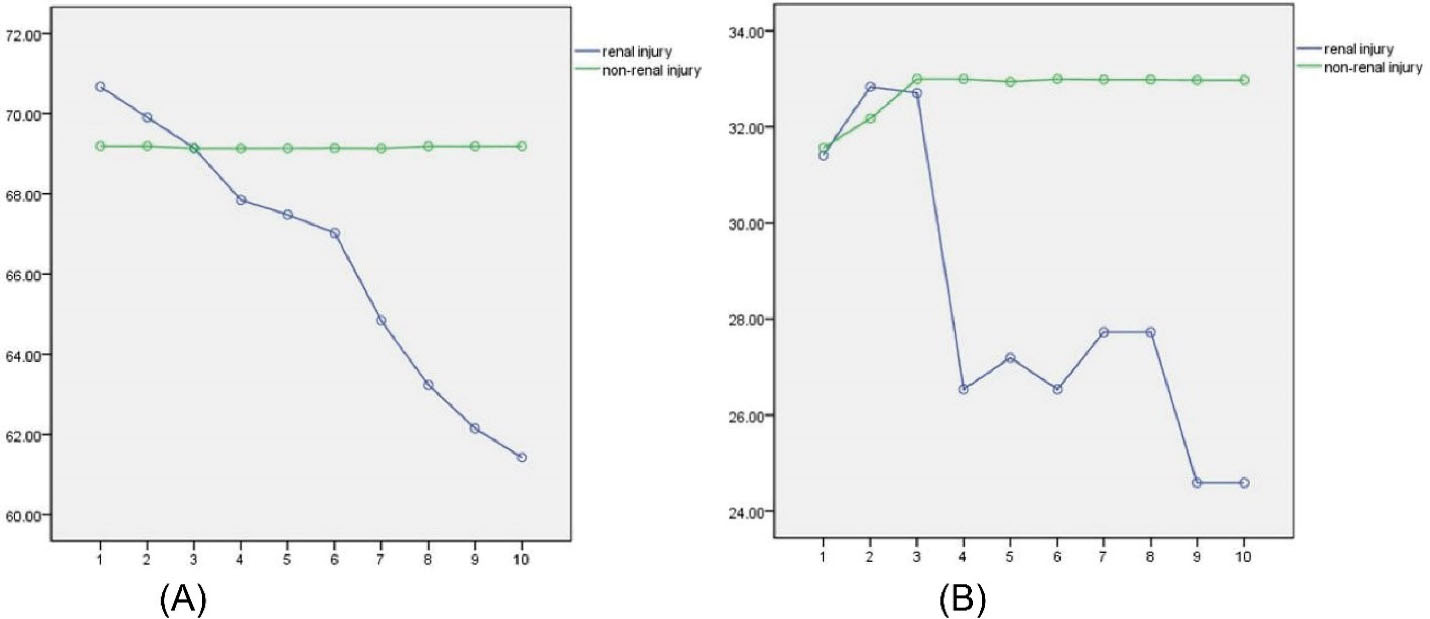
Figure 1.
(A) Changes in Renal Volume. (B) Changes in the Peak Systolic Velocity of the Renal Interlobar Artery.
.
(A) Changes in Renal Volume. (B) Changes in the Peak Systolic Velocity of the Renal Interlobar Artery.
The PSV of the renal interlobar artery in the renal injury group declined significantly after the third follow-up, and remained low. However, there were no significant changes in the non-renal injury group (Figure 1B). During follow-up, ultrasound played an important role in monitoring the changes in renal volume and intrarenal perfusion (Figure 2).
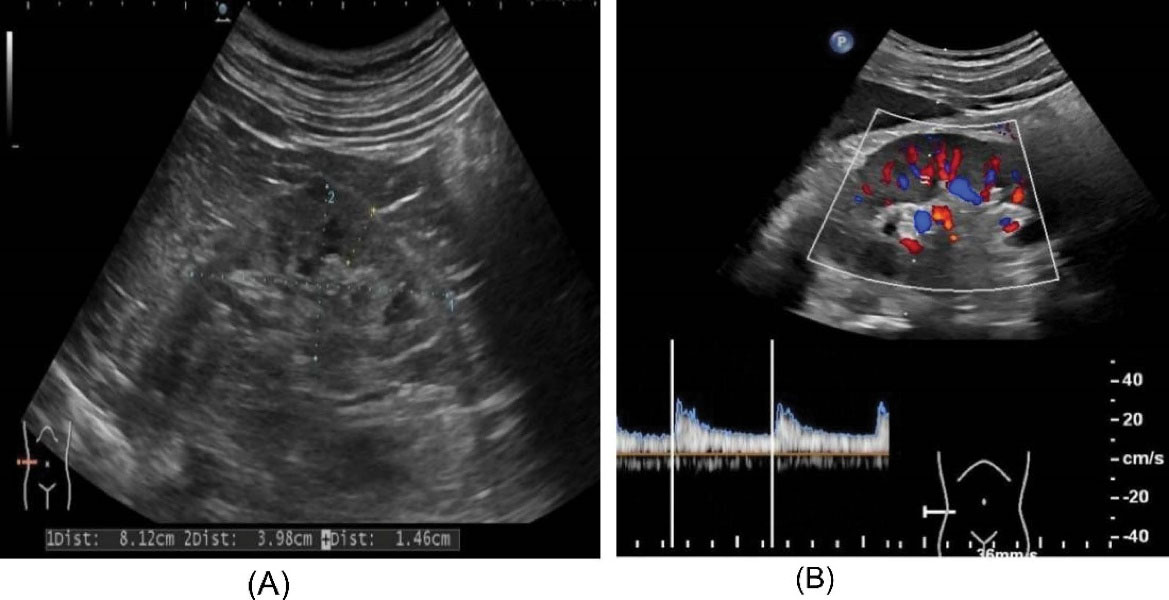
Figure 2.
Ultrasound Examination of the Kidney. (A) Ultrasound was used to monitor the condition of the insufficient kidneys and measure the renal volume in patients with kidney injury. (B) Ultrasound was used to detect the intrarenal perfusion and the peak systolic velocity of the renal interlobar artery in patients with renal injury.
.
Ultrasound Examination of the Kidney. (A) Ultrasound was used to monitor the condition of the insufficient kidneys and measure the renal volume in patients with kidney injury. (B) Ultrasound was used to detect the intrarenal perfusion and the peak systolic velocity of the renal interlobar artery in patients with renal injury.
Discussion
In this study, kidney cysts associated with renal injury were studied according to the characteristics of renal function and features of the renal cysts, including the number and maximum diameter of the cysts. SRCs were found to be associated with kidney injury, especially when patients presented with multiple renal cysts and cysts with a maximum diameter of more than 1.5 cm. In our study cohort, SRCs were mainly detected in male patients, and their size and number often increased with age. Male patients with multiple cysts of 1.5 cm or greater in diameter accounted for 73% of the renal injury group, but only 49.6% (P < 0.001) of the non-renal injury group. This is consistent with previous studies2 and suggests that the formation of cysts may be affected by the hormones different between males and females.
Analysis of the effects of SRCs on kidney damage, after adjustment for confounders, indicates that multiple cysts and cysts with a maximum diameter ≥ 1.5 cm were associated with renal injury. Marumo et al9 investigated this phenomenon. The results showed that renal cysts occurred and developed with epithelial cell proliferation, tubular dilatation and fluid retention. The pathological basis of renal cyst is mainly tubular obstruction and ischemia, so renal cyst occurs more frequently in the cortical area.
Our results revealed that patients in the renal injury group were significantly older than those in the non-renal injury group (57.7 vs. 47.5 years, P < 0.001). Progressive renal unit loss that gradually occurs with age may be important for the increased incidence of SRCs. Renal aging is characterized by the gradual appearance of renal atherosclerosis with the loss of normal functional glomeruli and the decline of overall renal function. To some extent, renal atherosclerosis causes a decrease in renal cortical volume. With advancing age, generally over 50 years of age, renal function begins to decline due to insufficient compensation of the residual renal unit. Renal vascular atherosclerosis and cortical volume loss are common pathophysiological phenomena in the elderly.10 Some researchers believe that age and hormonal changes play an important role in the formation of renal cysts.11 Ultrasound imaging can evaluate changes in renal morphology and renal blood perfusion, thus providing valuable information on changes in renal function.12 For chronic kidney disease (CKD), ultrasound evaluation is recommended to assess the longitudinal kidney diameter, parenchymal echo, and urinary tract conditions.13 Moreover, the longitudinal kidney diameter is considered an important marker of CKD as it decreases with the gradual decrease of GFR. This in turn reflects the decrease of renal function, accompanied by the reduction of renal volume, which is consistent with our results. In the comparison of the two groups, the renal volume decreased significantly in the renal injury group (P < 0.001), while some studies have shown that renal volume is a more accurate parameter reflecting renal function.14 In our report, the renal volume of patients in the renal injury group decreased slowly, reflecting the gradual decline of renal function over the follow-up period.
As for the change in the interlobular artery, color Doppler can analyze the renal blood flow perfusion and the change in blood flow velocity in renal blood vessels.15 Ultrasound monitoring of interlobar artery PSV is important for the assessment of renal artery status. During CKD, progressive glomerular atrophy and tubulointerstitial fibrosis are responsible for reduced renal blood flow.16 It is worth noting that these factors may also cause the formation of SRCs. As shown in Figure 1b, in the renal injury group, the PSV of the interlobular artery decreased significantly after the third follow-up, which suggests that renal blood perfusion will also decrease significantly during this period; however, the renal volume change manifested as a slow declining trend. Therefore, we inferred that in the kidney injury group, reduced renal perfusion occurred early and subsequently remained at a relatively low level, with hemodynamic changes further leading to reduced renal volume. No such changes were found in the non-renal injury group.
Furthermore, Plesiński et al17 reported that larger SRCs may increase the risk of hypertension. Larger SRCs compress the renal artery and activate the renin–angiotensin system, which is thought to be a possible mechanism by which larger renal cysts contribute to hypertension. Zerem and colleagues also noted that a gradual increase in cyst volume can lead to potential kidney disease or renal ischemia, which is an important cause of increased secondary renin secretion.18 As a result, the systolic and diastolic blood pressure in the renal injury group increased compared with the non-renal group, which is consistent with our observations. The age-related decline in the number of renal units may be related to hypertension, and elevated blood pressure is known to affect renal function.19 This may explain the interaction between the SRCs and renal injury. In our study cohort, hypertension, aging, male sex, obesity, smoking, and alcohol consumption were associated with SRCs, especially multiple cysts with a maximum diameter ≥ 1.5 cm.2,20 Identification of these characteristics provides an important theoretical basis for the prevention of SRC-related damage to kidney function.
There are several limitations to this study. First, our report did not examine the detailed mechanisms responsible for SRC formation, and this will require further molecular research. Second, although ultrasound examination is a reliable tool for estimating SRCs, it is highly dependent on operator proficiency. The evaluation of results is subjective, and it is easy to neglect smaller renal cysts. Third, this study was conducted in a racially homogenous cohort and further research will be needed to verify the results in a multi-racial cohort. Finally, the relatively small sample size of this investigation cannot be disregarded, and future research should focus on a larger sample size.
In conclusion, people with characteristics such as aging, obesity, smoking, drinking, and hypertension have a greater chance of suffering from renal cysts. Patients with larger multiple cysts are more susceptible to kidney injury. In patients with renal injury, a reduction in intrarenal perfusion was observed early, but the reduction in renal volume was a slow and progressive process. Ultrasound examinations can play an important role in monitoring renal volume and renal hemodynamic changes.
Authors’ Contribution
CL: Design and conception and data acquisition. MZ: Analysis and interpretation. CL and XC: Writing the manuscript. All authors critically revised the manuscript and gave final approval and agree to be accountable for all aspects of the paper.
Conflict of Interest Disclosures
The author has no conflicts of interest to declare.
Ethical Statement
Written informed consent was obtained from patients for publication of this article and any accompanying images. The author is accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
References
- Israel GM, Bosniak MA. An update of the Bosniak renal cyst classification system. Urology 2005; 66(3):484-8. doi: 10.1016/j.urology.2005.04.003 [Crossref] [ Google Scholar]
- Ozveren B, Onganer E, Türkeri LN. Simple renal cysts: prevalence, associated risk factors and follow-up in a health screening cohort. Urol J 2016; 13(1):2569-75. [ Google Scholar]
- Gimpel C, Avni EF, Breysem L, Burgmaier K, Caroli A, Cetiner M. Imaging of kidney cysts and cystic kidney diseases in children: an international working group consensus statement. Radiology 2019; 290(3):769-82. doi: 10.1148/radiol.2018181243 [Crossref] [ Google Scholar]
- Kwon T, Lim B, You D, Hong B, Hong JH, Kim CS. Simple renal cyst and renal dysfunction: a pilot study using dimercaptosuccinic acid renal scan. Nephrology (Carlton) 2016; 21(8):687-92. doi: 10.1111/nep.12654 [Crossref] [ Google Scholar]
- Hong S, Lim JH, Jeong IG, Choe J, Kim CS, Hong JH. What association exists between hypertension and simple renal cyst in a screened population?. J Hum Hypertens 2013; 27(9):539-44. doi: 10.1038/jhh.2013.12 [Crossref] [ Google Scholar]
- Chang CC, Kuo JY, Chan WL, Chen KK, Chang LS. Prevalence and clinical characteristics of simple renal cyst. J Chin Med Assoc 2007; 70(11):486-91. doi: 10.1016/s1726-4901(08)70046-7 [Crossref] [ Google Scholar]
- Park H, Kim CS. Natural 10-year history of simple renal cysts. Korean J Urol 2015; 56(5):351-6. doi: 10.4111/kju.2015.56.5.351 [Crossref] [ Google Scholar]
- Dorey TW, Moghtadaei M, Rose RA. Altered heart rate variability in angiotensin II-mediated hypertension is associated with impaired autonomic nervous system signaling and intrinsic sinoatrial node dysfunction. Heart Rhythm 2020; 17(8):1360-70. doi: 10.1016/j.hrthm.2020.03.014 [Crossref] [ Google Scholar]
- Marumo K, Horiguchi Y, Nakagawa K, Oya M, Ohigashi T, Asakura H. Incidence and growth pattern of simple cysts of the kidney in patients with asymptomatic microscopic hematuria. Int J Urol 2003; 10(2):63-7. doi: 10.1046/j.1442-2042.2003.00577.x [Crossref] [ Google Scholar]
- Daugirdas JT, Meyer K, Greene T, Butler RS, Poggio ED. Scaling of measured glomerular filtration rate in kidney donor candidates by anthropometric estimates of body surface area, body water, metabolic rate, or liver size. Clin J Am Soc Nephrol 2009; 4(10):1575-83. doi: 10.2215/cjn.05581008 [Crossref] [ Google Scholar]
- Simms RJ, Ong AC. How simple are ‘simple renal cysts’?. Nephrol Dial Transplant 2014; 29(Suppl 4):iv106-12. doi: 10.1093/ndt/gfu106 [Crossref] [ Google Scholar]
- Meola M, Samoni S, Petrucci I. Imaging in chronic kidney disease. Contrib Nephrol 2016; 188:69-80. doi: 10.1159/000445469 [Crossref] [ Google Scholar]
- Lucisano G, Comi N, Pelagi E, Cianfrone P, Fuiano L, Fuiano G. Can renal sonography be a reliable diagnostic tool in the assessment of chronic kidney disease?. J Ultrasound Med 2015; 34(2):299-306. doi: 10.7863/ultra.34.2.299 [Crossref] [ Google Scholar]
- Mansilla MA, Sompallae RR, Nishimura CJ, Kwitek AE, Kimble MJ, Freese ME. Targeted broad-based genetic testing by next-generation sequencing informs diagnosis and facilitates management in patients with kidney diseases. Nephrol Dial Transplant 2021; 36(2):295-305. doi: 10.1093/ndt/gfz173 [Crossref] [ Google Scholar]
- Zerem E, Imamović G, Omerović S. Simple renal cysts and arterial hypertension: does their evacuation decrease the blood pressure?. J Hypertens 2009; 27(10):2074-8. doi: 10.1097/HJH.0b013e32832f1458 [Crossref] [ Google Scholar]
- Ikee R, Kobayashi S, Hemmi N, Imakiire T, Kikuchi Y, Moriya H. Correlation between the resistive index by Doppler ultrasound and kidney function and histology. Am J Kidney Dis 2005; 46(4):603-9. doi: 10.1053/j.ajkd.2005.06.006 [Crossref] [ Google Scholar]
- Plesiński K, Adamczyk P, Świętochowska E, Morawiec-Knysak A, Gliwińska A, Korlacki W. Evaluation of liver-type fatty acid binding protein (L-FABP) and interleukin 6 in children with renal cysts. Adv Clin Exp Med 2019; 28(12):1675-82. doi: 10.17219/acem/110312 [Crossref] [ Google Scholar]
- Zerem E, Imamović G, Omerović S. Simple renal cysts and arterial hypertension: does their evacuation decrease the blood pressure?. J Hypertens 2009; 27(10):2074-8. doi: 10.1097/HJH.0b013e32832f1458 [Crossref] [ Google Scholar]
- Goto NA, van Loon IN, Morpey MI, Verhaar MC, Willems HC, Emmelot-Vonk MH. Geriatric assessment in elderly patients with end-stage kidney disease. Nephron 2019; 141(1):41-8. doi: 10.1159/000494222 [Crossref] [ Google Scholar]
- Han Y, Zhang M, Lu J, Zhang L, Han J, Zhao F. Hyperuricemia and overexcretion of uric acid increase the risk of simple renal cysts in type 2 diabetes. Sci Rep 2017; 7(1):3802. doi: 10.1038/s41598-017-04036-6 [Crossref] [ Google Scholar]